Services
Bioventix has created a novel sheep hybridoma technology to produce stable and productive cell lines that secrete sheep monoclonal antibodies (SMAs) of very high affinity. The ultra-high affinity of Bioventix’s antibodies facilitates the development of immunoassays of increased sensitivity and precision at Bioventix’s customers.
The technology to create monoclonal antibodies was first described in 1974 by Kohler & Millstein. Their work demonstrated that mouse cells could be created to make mouse monoclonal antibodies in large quantity.
Bioventix technology is related to this early work but focusses on the use of sheep cells to make sheep monoclonal antibodies (SMAs).
Bioventix will take your immunogen or assist with the design of an appropriate immunogen for immunisation into sheep. Test bleeds are taken and ELISAs performed to confirm the response of the sheep to the immunogens.
Multiple hybridoma fusions are performed yielding cell culture supernatants that can be assayed for their reactivity by additional ELISA screens set up together with the customer. A choice then can be made of clones worthy of further cloning and scale-up to yield quantities of purified SMAs for evaluation back at the customers laboratory.
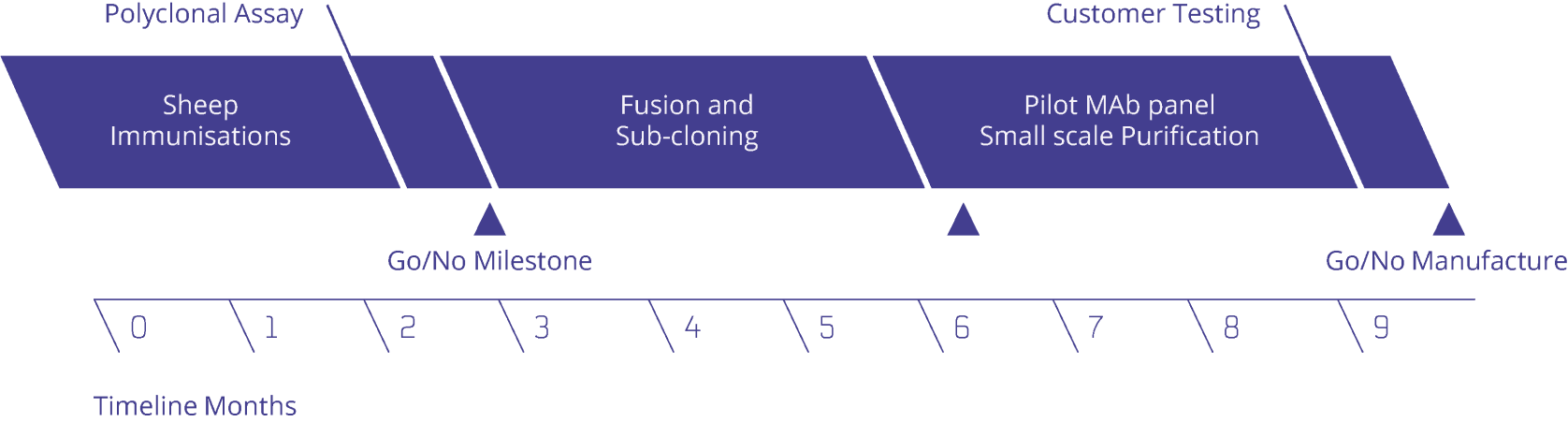
Scroll to see more
The process operates in a stepwise structure with customer authorisations to proceed to the next step at each milestone: pre-fusion screens, sub-clone analysis and pilot scale-up. The entire process takes around nine months to complete. Please contact us for further details.

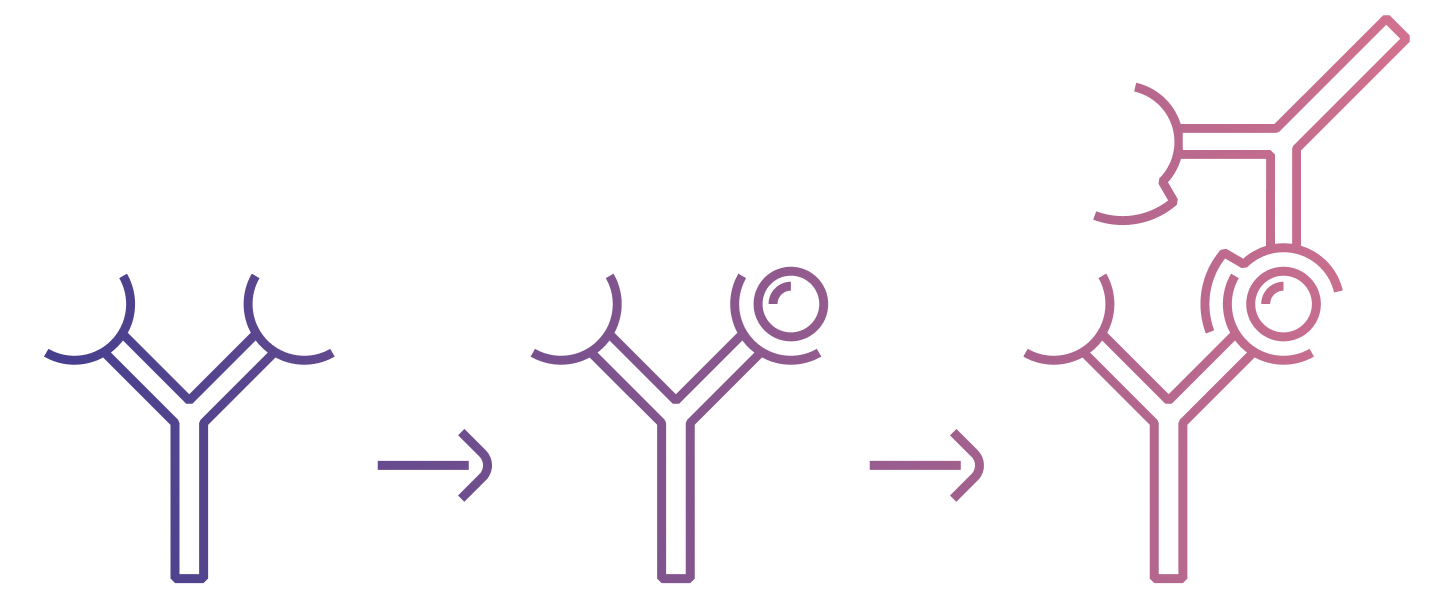
In immunoassays, the ‘sandwich’ format involves two antibodies working in tandem to bind to different epitopes (binding sites) of the same analyte. The process begins with a primary antibody binding to the analyte, capturing it on its surface. Following this, a secondary antibody binds to a different epitope of the same analyte. This results in the analyte being ‘sandwiched’ between the two antibodies.
The result of a sandwich assay is directly proportional to the concentration of the analyte. This is a significant advantage over the competitive assay format, which only uses a single analyte specific antibody. The use of two antibodies in the sandwich assay enhances both specificity and sensitivity, making it a preferred choice for many applications. However, it’s important to note that sandwich assays may not be suitable for all types of analytes. Specifically, small molecule analytes that lack two accessible immunogenic epitopes cannot be detected using this method.
Unlike the secondary antibody in a traditional sandwich assay, an anti-complex antibody doesn’t bind directly to the analyte. Instead, it binds to both the analyte and the primary antibody when they are in a bound state, known as an immune complex. Since the anti-complex antibody doesn’t need to bind directly to the analyte, it opens the possibility of ‘sandwiching’ small molecule targets. This is a significant advantage, as small molecules often present challenges in traditional immunoassays due to their size and lack of multiple epitopes.
We are always delighted to help customers and collaborative partners learn more about how Bioventix can elevate your testing capabilities.
Please contact us to experience first-hand the enhanced performance in lateral flow assays for targets like THC and cortisol with Bioventix high-affinity SMAs and their highly specific anti-complex sandwich counterparts.